The occurrence of AGE is caused by the constant presence of various sugars in the blood (for example, glucose, fructose and lactose), which cause glycation processes in the tissues of our body. The active consumption of sugars with food accelerates the formation of AGE. At the same time, some food products contain AGE in finished form. They are formed as a result of thermal processing of food (above 120°C): when frying or baking. A crust on bread or kebabs is the AGE. In addition, AGEs can be generated from UV radiation (especially in the skin) and tobacco smoke.
The negative effects of AGEs are carried out in two main ways. First, they form cross-links between long-lived molecules, which is described in detail in the section “Matrix aging and glycation”. Secondly, they interact with their main receptor located on the cell surface—RAGE (Receptor for Advanced Glycation End products), triggering an intracellular cascade of events leading to the activation of a number of inflammation genes.
RAGE receptors
RAGE receptors belong to the immunoglobulin family. They are activated by AGE, as well as amyloids and cell death products. The highest content of RAGE receptors is observed in the lungs; in addition, they are widely distributed in adipose tissue and are involved in the regulation of insulin signaling. There is a hypothesis that the original role of hominid RAGE receptors was associated with the formation of an economical phenotype with accelerated fat gains, which saved us from starvation.RAGEs are now known to be involved in nearly all age-related conditions, including both types of diabetes, chronic kidney disease, cardiovascular disease, cancer, and Alzheimer's and Parkinson's. In response to the binding of the RAGE receptor, the transcription factor NF-κB is activated, which “turns on” (triggers the expression) of a number of genes associated with inflammation. In addition, it further activates RAGE, enhancing inflammation in a positive feedback loop. RAGE activation also leads to the rapid generation of reactive oxygen species (ROS).
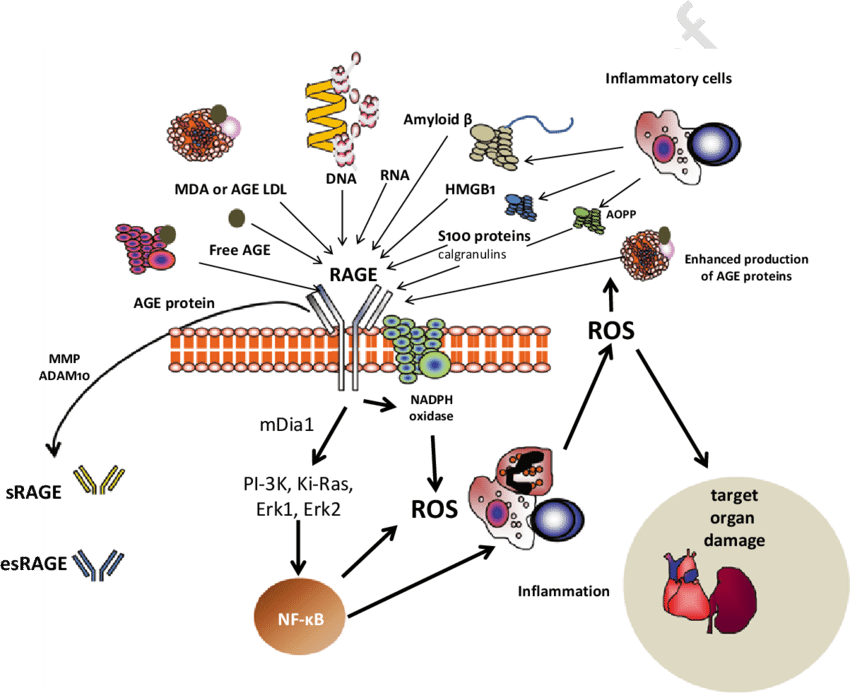
RAGE receptors are present in our body in two forms: membrane bound mRAGE and soluble sRAGE. They differ in structure: mRAGE has three domains, while sRAGE contains only one, extracellular domain.
Soluble RAGE Form
It is known that in response to the binding of RAGE receptors by end products of glycation, cells begin to produce a soluble form - sRAGE. Therefore, it serves as a biomarker of AGE-induced inflammation. It is also able to compete for the binding of AGE with the RAGE receptor on membrane surfaces and thus slow down the inflammation process. In this regard, sRAGE is used as a basis for the development of anti-inflammatory agents. For example, Japanese researchers have developed hybrid protein "traps" for AGE, consisting of a region of the RAGE receptor that binds AGE and an elastin-like protein.Such protein-engineered products already have a future as anti-inflammatory, wound-healing and geroprotective drugs.
For the same purpose, you can use non-protein agents with a lower molecular weight. For example,DNA aptamers are oligonucleotides aimed at blocking RAGE receptors that bind matrix proteins to AGE. They are planned to be used to restore organs damaged due to glycation.
The ability to block RAGE receptors has been found in some artificially created amino acids, in deuterated polyunsaturated fatty acids, chebulic acid, quercetin, and the GLY-230 molecule. The main disadvantage of these molecules is that in low concentrations, AGE blockers are ineffective, and in high concentrations, they are toxic. The system, however, can be changed by selecting new, more effective and safer AGE blockers.
